A) it is rarely needed
B) the high pressure makes it impossible
C) the reaction occurs to fast
D) it breaks down in the extreme heat
E) none of the above
G) None of the above
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Mercury fulminate is a shock-sensitive primary explosive that was used in primers in firearms ammunition. It has an OB% of -11.2. Suppose it is combined with potassium perchlorate (OB +46.2) in a formulation. What mixture would yield a net zero oxygen balance? Report in %mercury fulminate/% potassium perchlorate
A) 73/27
B) 48/52
C) 81/19
D) 61/39
E) none of the above
G) B) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
The explosive ethylenedinitramine has a formula weight of 150. g/mole and a heat of explosion of 4278 kJ/kg. What is the heat of explosion in kJ/mole?
A) 29
B) 129
C) 824
D) 642
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is a pipe bomb that is made with steel typically more powerful than one made with plastic pipe?
A) steel is more malleable than plastic
B) often the plastic won't rupture at all
C) greater pressure can build in the device before it fails
D) steel doesn't get as hot as plastic
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ability of an explosive to cause fragmentation and shrapnel formation is called
A) brisance
B) pushing power
C) relative power
D) QV
E) none of the above
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following information:
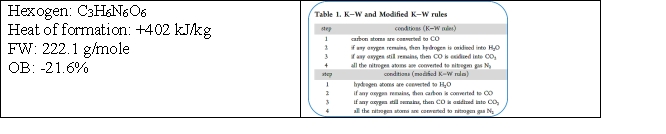 How many moles of gas are produced by the explosion of 1 mole of hexogen?
How many moles of gas are produced by the explosion of 1 mole of hexogen?
A) 3
B) 5
C) 7
D) 9
E) none of the above
G) D) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
What is the difference between a detonation and an explosion?
A) detonation differs from simple combustion in the speed of the reaction and pressure
B) nothing; they mean the same thing
C) An explosion can only be caused by detonation
D) A detonation can only be caused by an explosion
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following information: 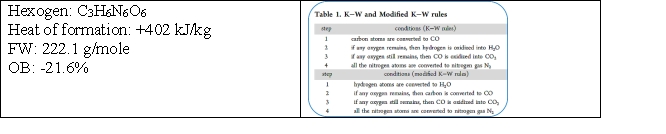 Predict the reaction products using the appropriate rules. How many moles of CO2 are produced in an explosion type reaction of 1 mole of hexogen?
Predict the reaction products using the appropriate rules. How many moles of CO2 are produced in an explosion type reaction of 1 mole of hexogen?
A) 1
B) 2
C) 3
D) 4
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the order of explosives built into an explosive train detonator?
A) least sensitive to most
B) most sensitive to least
C) least-most-least
D) most-least-most
E) none of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Pull-poppers are small devices that detonate with a loud pop if stepped on. This would be classified as a
A) secondary tertiary explosive
B) secondary low explosive
C) primary high explosive
D) military explosive
E) none of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 10 of 10
Related Exams